From cortisone to CAT scans, the future of medicine has often arrived first at the Mayo Clinic. The tradition continues today with the fit-up of a new headquarters for regenerative biotherapeutics adjacent to the research center’s Rochester, Minn., campus. The fourth floor of the recently built Discovery Square building, Two Discovery Square, had been earmarked originally for office space, but after COVID hit, the developer offered the opportunity to convert some of the office space to laboratory space. Mayo Clinic had been planning to build a Center for Regenerative Biotherapeutics and saw an opportunity to lease the 18,000-sf space at Discovery Square and convert it for this cutting-edge biomanufacturing facility.
Regenerative biotherapeutics is a promising new frontier in health science. A regenerative biotherapeutic treatment takes human or animal cells and tissues and reengineers them to perform a new job, such as reprogramming T-cells to attack specific cancers. “We often use the term ‘healing from within,’” says Henry Walker, senior program manager for Mayo Clinic.
Before the new facility opened in mid-2024, many of these innovations followed a necessarily long process from drug discovery to development before reaching the patient. Speeding up that pace was an important goal for the clinic. Mayo researchers “needed to be able to get these projects to manufacturing faster, but they didn’t have the spaces that supported that, or the space to support the quality control that was needed,” says Chris Stenzel, project manager for facilities project services at Mayo.
Rethinking Discovery Square
The aim of the fourth-floor fit-up was to widen that bottleneck by accelerating the pace of experimental therapies through first- and second-phase clinical trials, according to Walker.
Mayo project stakeholders saw a number of advantages in repurposing a portion of the Discovery building for biotherapeutics. First and foremost was the location adjacent to the main campus. It also allowed Mayo to consolidate quality control operations that support the Center for Regenerative Biotherapeutics. Mayo did not disclose the cost of the project but noted that there were cost advantages to leasing Two Discovery, which is owned by outside investors on Mayo-owned property.
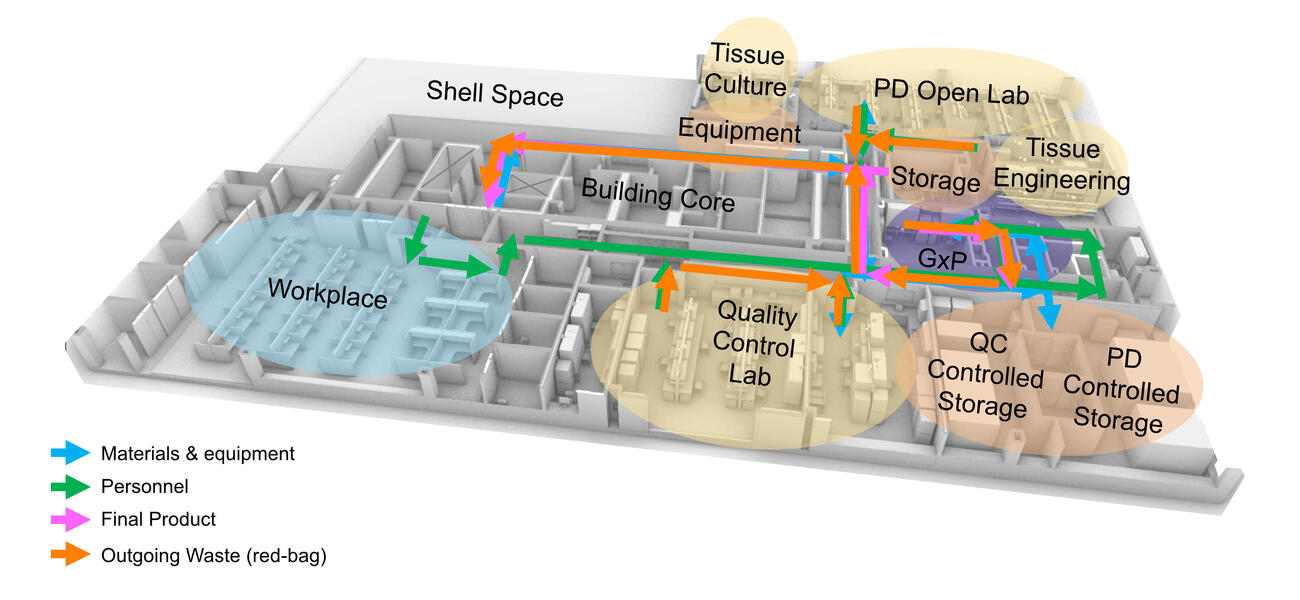
Designed by a team from Flad Architects’ Madison, Wis., office, the floor establishes a new biomanufacturing facility for process development alongside GxP, quality assurance, and phase 1 and 2 trials. GxP is an umbrella term for which the x refers to regulatory-compliant spaces such as Good Manufacturing Practice, Good Laboratory Practice, Good Clinical Practice for clinical trials, or Good Documentation Practice for documenting distribution practices for new therapies. Establishing a baseline of regulatory compliance allows for improved efficiency and flexibility as the space needs shift for each new research initiative and from research to manufacturing.
“I can’t emphasize enough how key the GxP has become to breaking the pipeline logjam to opening patient delivery,” says Walker. “It’s a game changer.”
Within the GxP lab, Mayo installed HVAC capacity with an air turnover rate of over 70 air changes per hour, which is at the high end of the range for a similar facility. Adding more people to a space increases particle counts, so a high air-change rate maximizes the space’s flexibility to accommodate more high-particle-producing procedures as well as more researchers.
Other institutions might have outsourced their environmental monitoring of the GxP suites, but Walker notes that Mayo decided to keep that function in house. “The FDA expects us to know and understand our facilities, including nuances, to have better control over our manufacturing processes,” he says. “By implementing a full monitoring program within our GxP space, we are ready to understand the nuances of a new product, or onboard an existing product into the space efficiently to help a patient in need.”
Plenty of interior windows into each lab provide good lines of sight and natural light throughout the spaces. “Along with the exterior windows, which have been shown to improve wellbeing and support a connection to nature, the interior windows provide a great deal of daylighting, which creates an inviting environment, enhances safety and productivity, and contributes to the overall health and wellbeing of the users,” says Sarah Jenne, project manager for Flad.
Additionally interior windows improve safety, oversight, and collaboration. They also offer visitors a view into the work being done, a consideration for regulatory inspection and tour routes for potential investors, who have been inspired by seeing science on display.
Additional spaces include:
Process Development – An open “ballroom”-style lab that can accommodate about 22 people and up to six projects at a time, each of which may require anywhere from two months to two years.
The PD facility also was built with flexibility in mind, to handle the differing requirements of the 100 or more projects that might be in the queue at any one time.
Flad proposed many flexible design features to facilitate swapping out equipment, such as mobile instrument carts in the center of the labs, minimal fixed casework, and overhead service panels that have a plug-and-play approach for power, data, and gases. The lab’s air-handling system is also designed with added capacity to evacuate additional heat generated by future equipment.
Efficient operational flows and the strategic alignment of lab adjacencies were key drivers in the planning of this project. The PD lab was located near the GxP suite to enhance workflow efficiency, foster collaboration among project teams, and mitigate the risk of cross-contamination. “We placed a strong emphasis on optimizing operational flows and lab adjacencies to not only improve efficiency but also actively reduce contamination risk,” says Cole Brunson, principal planner and process architect for Flad.
Quality control – Consolidating these testing labs aligns procedures and techniques to leverage best testing outcomes. Now, instead of multiple labs needing to qualify the same test outcome, one testing lab qualifies one test process. As validation is time-consuming and costly, one lab can do this far more efficiently than multiple labs.
Lessons learned
Looking back at the project, planners and designers came away with several important lessons about how to fit out an ordinary building in a high-end research facility:
- Expect the unexpected. When adapting a building for a new use – even a relatively new building – there will usually be surprises that may not be dealbreakers but will need to be addressed. It’s helpful to leverage design expertise and carefully consider required building criteria before negotiating lease terms. Understand the building and infrastructure’s current state and what will need to be adapted.
“If you are not building from the ground up,” warns Jenne, “there are many things to consider for a complex scientific space like this, including the shell, structural load and utility capacity, vibration, and more.”
“These spaces have to be very, very highly controlled for temperature and humidity, and whatever needs to be done to keep out insects and things like that,” adds Stenzel.
In the case of Two Discovery Square, the structural limitations weren’t immediately apparent. “The exterior walls, on a plan in architectural world and CAD (computer-aided design) world, looked great,” says Stenzel. “Everything was perfect. You get into the actuals of it, and you find that this wall wasn’t quite where it was supposed to be; it was bumped out. So, instead of being able to have a straight wall, you would have had to do all these jigs and jogs along the wall. We worked with Flad and our contracting team to come up with some ways to negotiate around those things.”
- Stay flexible. Planning a facility where the research will be constantly evolving, Mayo wanted to ensure they thought of everything. “I asked for a lot of power, extra power,” says Walker. “I asked for a lot of redundancy. If there’s one line for compressed gas, I need an N+1 system so I have backup for everything, and I need extra circuits for the panels, because all the equipment is very sensitive and needs its own circuit.”
- Talk with the experts. “How do you get your air, your large ducting through and still make that GxP space function when it takes up a ton of space in the upper deck?” says Walker. “Building information modeling can tell you a lot about that, but so can the subcontractors who have done this for many years. They can look at it and say, ‘We can’t get our ducting through here. We have to make changes somewhere, somehow.’”
Conversations with their vendors had other uses as well. “Chris Stenzel was really good about engaging all these vendors and getting them on board, doing the planning ahead of time and helping them understand the goal isn’t just four walls,” says Walker. “Once they understand that their work correlates directly to the outcome of a patient, you have buy-in.”
By Bennett Voyles